Product Description
Genomic Cancer Panel and Profiling Markets by Cancer, Application, Tissue and Gene Type with Screening Potential Market Size and Executive/Consultant Guides 2023 to 2027
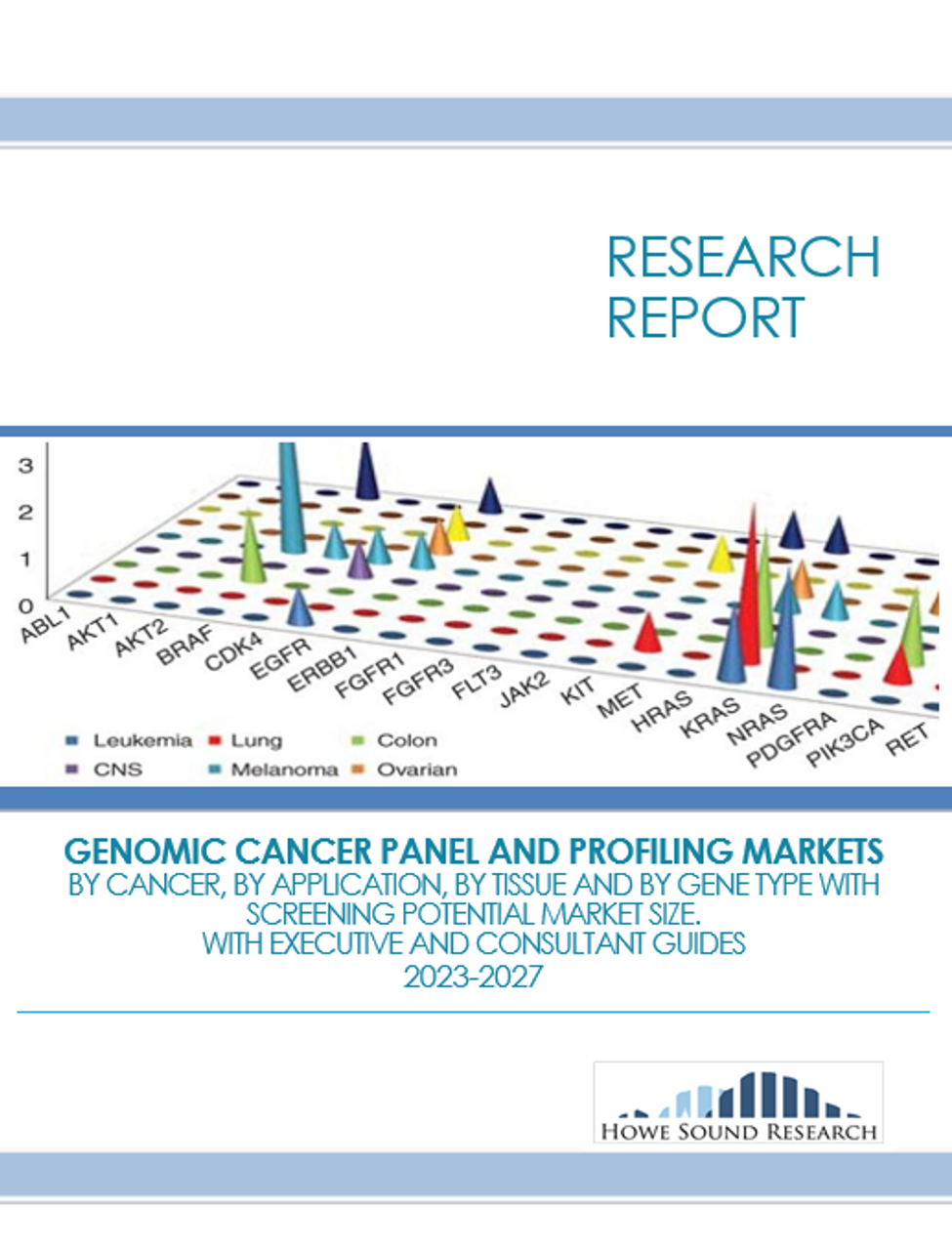
“Cancer diagnosis and treatment is entering a new era. Large Profile Panels are dominating the market. Clinicians and Pharmaceutical customers building global demand and a new diagnostic industry based on Cancer Gene Sequencing. What role will Whole Genome Sequencing eventually play?”
Publication date: September 2023
Page count: 591
Cancer gene panels and genomic profiling are quickly changing the diagnosis and treatment of cancers. The market is moving out of a specialized niche and going mainstream as Oncologists begin routinely using information on the hundreds of genes related to cancer. The market is exploding as physicians use all the information they can get in the battle against cancer while pharmaceutical companies see the potential to make nearly any therapy viable.
This comprehensive report includes data on how test volumes have grown for the biggest players. Find out how this new way of understanding cancer will change cancer diagnostics forever.
Comprehensive panels, genomic profiling, high risk breast cancer panels. Learn all about how players are jockeying for position in a market that is being created from scratch, with some players pulling way out in front and expanding globally. This is a dynamic market situation with enormous opportunity where the right diagnostic with the correct support can command premium pricing. The science is developing whilst at the same time creating new opportunities with regularity... and the cost of sequencing continues to fall.
The report includes a complete list of Current 2022 United States Medicare Fee Payment Schedules to help understand the new pricing for cancer panels. Forecast demand for new testing regimes or technologies. Make research investment decisions.
ABOUT THE LEAD AUTHOR
Mr. Greg Powell, B.SC., M.B.A is an experienced business and clinical professional. He is co-author of the paper "The Radioimmunoassay of Angiotensinogen by Antibody Trapping." He has worked in laboratory testing and management for over 20 years. Mr. Powell’s education includes:
- B.Sc. (Chemistry) University of BC
- M.B.A. (Finance and Policy) University of BC
- Market Research Seminar - Burke Institute
- Finance for Senior Executives - Harvard Business School